Carbon Sequestration
by Andrew Boyd
Today, the world's highest profile molecule. The University of Houston's College of Engineering presents this series about the machines that make our civilization run, and the people whose ingenuity created them.
The air we breathe is a mixture of many different gases. Some, known as greenhouse gasses, trap the sun's heat. And that's a good thing. Without any greenhouse gasses the earth's surface would freeze.
But it's possible to have too much of a good thing. Carbon dioxide is an important greenhouse gas used by plants to create energy and oxygen. We create carbon dioxide in the process of breathing. But carbon dioxide is also created when we burn coal, oil, and natural gas — our major sources of power. The long and divisive debate about global warming stems from the carbon dioxide released when we burn these fossil fuels.

So engineers have asked what can be done to capture carbon dioxide and keep it out of the atmosphere. It's actually one of fourteen grand challenges for the twenty-first century put forth by the National Academy of Engineering. And it's receiving a lot of attention and research dollars.
One of many challenging questions is what to do with the carbon dioxide once we've captured it. And here's where engineers are displaying their creativity.
Among the possibilities are efforts to convert it to something else — a project chemical engineers are working on. One idea focuses on creating a chemical that could power electric cars. Another novel idea involves embedding carbon dioxide in plastics. Agricultural engineers are keeping busy, too. Their ideas range from the simple — grow more green plants — to the advanced — engineer plants that use more carbon dioxide. Oceanographic engineers are looking to the seas — pumping liquid carbon dioxide to depths where it's denser than sea water and sinks.

But perhaps the most promising method is simply pumping unwanted carbon dioxide underground. It sounds a bit horrifying at first, conjuring images of dangerous bubbles waiting to explode onto the surface. But that's the wrong image.
Carbon dioxide isn't in itself hazardous to life. It's what makes Coke, Pepsi, and other soft drinks fizzy. It's an important part of the carbon cycle necessary for life. What makes it a concern is the greenhouse effect from having too much of it in the air.
We've already created empty space underground by removing fossil fuels. Depleted oil and gas wells are natural candidates for storing the unwanted gas. In some cases, the carbon dioxide can actually be used to push extra oil or natural gas out of existing reservoirs.
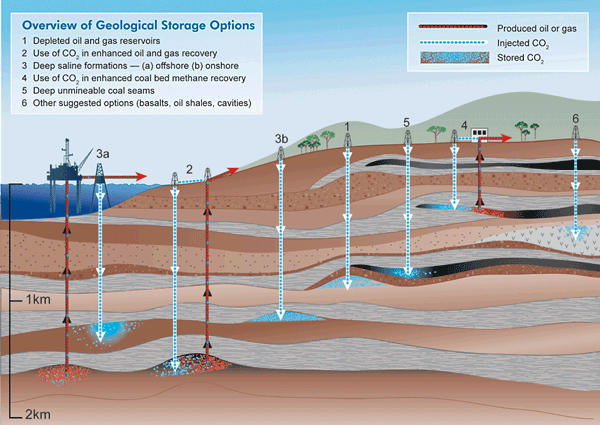
Are these the best ways to treat unwanted carbon dioxide? Or should we stop creating it by reducing our consumption of fossil fuels? Or for that matter, should we be worrying about it so much? Only one thing's for certain: they're all good engineering questions.
I'm Andy Boyd at the University of Houston, where we're interested in the way inventive minds work.
Notes and references:
Carbon Sequestration. From the U.S. Department of Energy website: http://www.fossil.energy.gov/programs/sequestration/index.html. Accessed August 28, 2012.
Develop Carbon Sequestration Methods. From the National Academy of Engineering Grand Challenges website: http://www.engineeringchallenges.org/cms/8996/9077.aspx. Accessed August 28, 2012.
Fossil Energy. From the U.S. Department of Energy website: http://www.fossil.energy.gov/programs/sequestration/geologic/index.html. Accessed August 28, 2012.
Greenhouse — Green Planet. From the PBS website: http://www.pbs.org/wgbh/nova/ice/greenhouse.html. Accessed August 28, 2012.
S. Ritter. 'What Can We Do With Carbon Dioxide?' Chemical and Engineering News, April 30, 2007. See also: http://pubs.acs.org/cen/coverstory/85/8518cover.html. Accessed August 28, 2012.
The picture of the smoke is by John Lienhard. All other pictures are from U.S. government websites.
This episode was first aired on August 30, 2012