Entropy at the Movies
Today, entropy at the movies. The University of Houston's College of Engineering presents this series about the machines that make our civilization run, and the people whose ingenuity created them.
I have huge admiration for film-critic Roger Ebert -- the way he combines fine writing with uncanny insight into the meaning of movies. And his love of the medium is contagious. Still, he's done two things that really bother me: One was giving four stars to that atrocious misogynistic movie, The Joy Luck Club. Well, we all blow one now and then -- and there's no settling arguments over taste.
Ebert's greater mistake was a line in his review of the movie, The Adjustment Bureau. In it, he says, "... in the long run the universe will entropy and cease." It'll do what!
Richard Armstrong recently did an Engines episode on the verbing of nouns -- that is, our rising inclination to make nouns into verbs: Text becomes texting; friend becomes friending, and so on. Now Ebert has verbed the noun entropy. And that makes it very hard on those of us charged with using and explaining science.
You see, entropy is a material property -- like density or elasticity. When we do things to a material we change its entropy. That much would be straightforward. But entropy seems mysterious for two reasons. One is that we can't measure it; we can only calculate it from other properties. We can't just examine two objects and know which of the two has the greater entropy.
It's also perplexing because, when anything happens, the combined entropy of everything involved in the event has to increase. The entropy of any object can increase or decrease. But together, the sum of all entropies is higher when the process is finished. That fact is called the second law of thermodynamics. It gives time its direction since time and entropy increase together. The changing entropy of all things defines past and present.
So you see the problem. Here's a concrete physical property that we engineers use when we design heat pumps or chemical processes, engines or heating systems. But it so happens that throughout the universe the sum of all entropies can only increase.
Try this: Put a liter of cold water in the refrigerator and let it freeze. After it's frozen, its entropy -- that technical noun -- will've decreased by around 1.3 kilojoules per degree Kelvin. Of course the entropy of the air around it will've increased. The point is, component entropies can change either way.
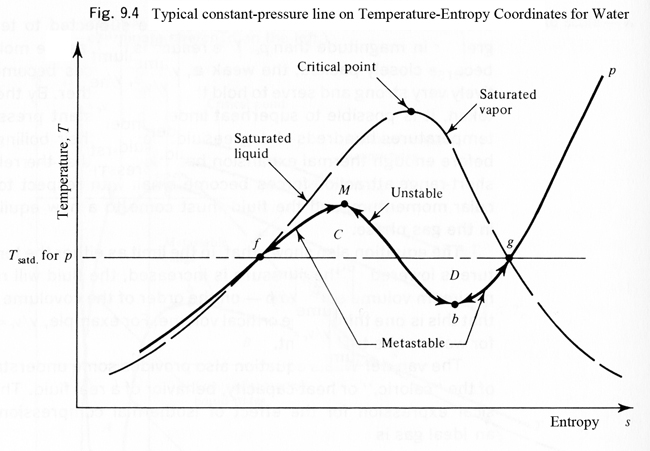
A schematic diagram showing how temperature varies with entropy for a typical fluid. The dome represents the liquid and vapor states at the boiling/condensing condition as pressure is varied. One typical line of constant pressure is also shown. (Image from Tien and Lienhard Statistical Thermodynamics.)
So we have the second law, with its cosmic implications of decay. But let's not steal entropy -- a working tool of engineering -- and make it into a verb meaning second law decay.
We need entropy in our work. And it'll be an impoverished world if we have to develop separate vocabularies -- one for sensible science and one for media hyperbole. To you, Mr. Ebert, I say thank you for years of enriching my movie-going. I'll even pretend I never saw that Joy Luck Club review. But please -- tread carefully on the precise language of engineering. For it is the coin by which we all negotiate the complex world we live in.
I'm John Lienhard at the University of Houston, where we're interested in the way inventive minds work.
(Theme music)
See the Wikipedia articles on The Second Law of Thermodynamics and Entropy. See also the IMDB article on The Joy Luck Club, as well as Armstrong's episode on verbing and Lienhard's episodes on the second law of thermodynamics and on teaching thermodynamics. My thanks to engineering colleague Keith Hollingsworth for his counsel.

The second law of thermodynamics tells us that things happen irreversibly -- that no real occurrence can ever be completely undone. (Clipart image from 19th-century source.)