Renewable Energy
Today, a neat way to get energy. The University of Houston's College of Engineering presents this series about the machines that make our civilization run, and the peoplewhose ingenuity created them.
It's a beautiful sunny day. The bright green leaves are dancing with the light breeze. We're driving on a long road toward our destination. The fuel dial shows we're low. We stop at the nearest fuel station to refuel. Among the fuel selections, we notice an extra option: Hydrogen. This scenario is actually coming to life. Yes, Hydrogen fuel cell electric cars are becoming a reality.
Hydrogen fuel cells offer many attractions. They use renewable energy sources like hydrogen and air. They're very friendly to our environment. But there's more.
They also produce clean water. That's why they're popular on space shuttles — two major benefits with only one device.

Hydrogen fueling nozzle [Wikipedia/EREE]
Let's take a peek into the fuel cell and unveil its mystery: We see the hydrogen and oxygen come in from two different sides. Both are ionized through special chemical reactions. In other words, the hydrogen gives away and the oxygen receives electrons.
Next, the hydrogen ions move through a special plastic material. They reach the other side where the oxygen ions are waiting. The hydrogen and oxygen ions combine and produce water. What happens to the stripped away electrons from the hydrogen? They flow through an external circuit and produce electricity.
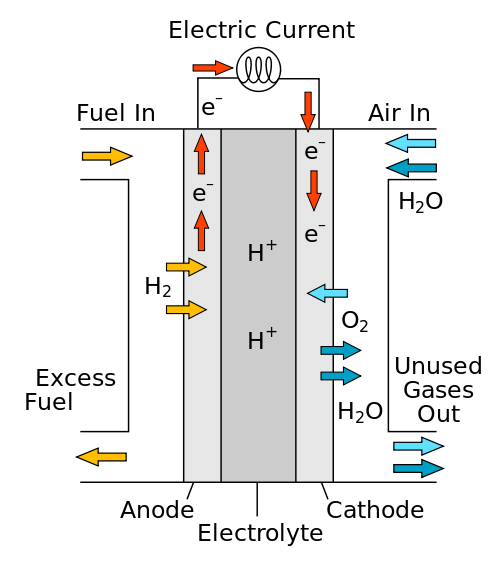
Hydrogen fuel cell [Wikipedia/R.Dervisoglu]
Many of us are familiar with lithium ion batteries. We use them in our laptops, cell phones, watches and hybrid cars. The hydrogen fuel cell and lithium ion battery are similar in many ways. They both produce electricity and offer renewable energy. But, even their inner workings have similarities: They both have two electrodes, an electrolyte and a separator. They both have a positive ion moving from one electrode to the other.
They're also different in some ways: The hydrogen fuel cell is an open system meaning that the fuel is replenished continuously. The lithium ion battery is a closed system and the materials are not resupplied. A fuel cell can't be recharged while a lithium ion battery is rechargeable. The lithium ion in the battery moves back and forth during charge and discharge cycles. The hydrogen ion in the fuel cell moves in one direction only.
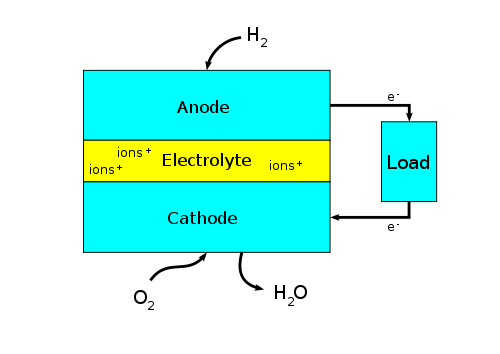
Block diagram of a fuel cell. [Wikipedia/Paulsmith99]
General Electric made the first hydrogen fuel cell in the 1960s. But problems of efficiency and cost prevented their commercialization for a long time. Engineers and scientists have tackled these problems using new and advanced materials. And perhaps some of us may soon press the hydrogen selection button to refuel our car.
I'm Haleh Ardebili at the University of Houston, where we're interested in the way inventive minds work.
(Theme music)
A hydrogen fuel cell is also called a "proton exchange membrane fuel cell" or PEMFC. Let's think about the hydrogen atom. Imagine one negative electron circling around a positive proton center. When we strip away the electron from the hydrogen, what remains is simply a "proton."
The special plastic material that separates the hydrogen and oxygen ions in the fuel cell is called a "proton exchange membrane" or PEM. Some people also call this membrane a "polymer electrolyte membrane." The hydrogen ion or proton must travel through PEM to reach the other side and combine with the oxygen ions and produce water.
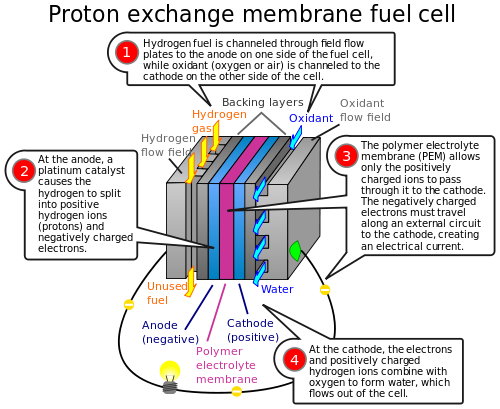
Proton exchange membrane fuel cell [Wikipedia/Kpengboy]
This episode first aired on October 22, 2013.