When Metal Grows Tired
Today, metal grows tired. The University of Houston's College of Engineering presents this series about the machines that make our civilization run, and the people whose ingenuity created them.
Remember the 1988 Aloha Airlines explosion? A metal fatigue failure - decompression blew a huge hole in the body - scores of hurt passengers; a stewardess died.
Metal fatigue is serious business, so let's ask how it works. Our understanding of it has changed since I studied engineering in the 1940s. Back then, we knew we could subject a metal part to an oscillating stress indefinitely only if we kept the stresses low. Certainly, if we deform it, we're in trouble. We've all bent a paper clip or a coat hanger. Bend either one slightly, and it'll come back to its original shape. Bend it far enough to deform it, then repeat the bending several times, and it'll break.
So think about cars, planes, bridges, and buildings. Their stresses can go through millions of cycles. In the 1940s, engineers ran long tests on machines that bent samples back and forth. The breaking stress fell off as the logarithm of the number of cycles. When the cycled stress was low enough, the part would never break.
Now the catch: we know that, if we cycle a metal through stresses high enough to permanently deform it, it'll soon break. But a metal subjected to many stress cycles seemingly fails at a stress too low to cause permanent deformation.
And we know something we didn't know back then -- that fatigue failures begin from tiny cracks -- maybe a micron (or a thousand nanometers) long. It still takes permanent deformation to make a metal fail in fatigue. But it occurs around those tiny cracks, while the gross behavior of the part still seems to be elastic. Now we have to ask whether the deformation caused by such cracks will make them propagate or stop. That changes the game: We must now find the level of stress that still allows the molecular structure around these invisible cracks to resist spreading.
Those micro-cracks usually gain a foothold around what we call stress concentrations -- where the peculiar shape of a stressed part boosts the stress. Suppose a wide metal strap carries a heavy load. Now drill a small rivet hole through the middle. The stress on either side of the hole is three times what it would be without the hole. That's where we look for trouble. Here's an example:
 In 1952, the deHavilland Comet, became the first jet airliner to carry passengers. Then in Spring, 1954, two Comets inexplicably crashed out of clear sky. Engineers finally learned that the window frames had suffered fatigue failures around rivet holes. As Comets took off and landed, pressurized and depressurized, they'd undergone about three thousand stress cycles before they failed.
In 1952, the deHavilland Comet, became the first jet airliner to carry passengers. Then in Spring, 1954, two Comets inexplicably crashed out of clear sky. Engineers finally learned that the window frames had suffered fatigue failures around rivet holes. As Comets took off and landed, pressurized and depressurized, they'd undergone about three thousand stress cycles before they failed.
Well, we're a lot better at anticipating that kind of failure today. But the game is far from over, especially since the cost of failure can be so high. It is odd, is it not, that, as we build ever-larger structures, our engineering focus is drawn down into the tiniest corners of the materials from which those structures are made.
I'm John Lienhard, at the University of Houston, where we're interested in the way inventive minds work.
For three snapshots of our understanding of fatigue one might look at books on the subject from 1941, 1968, and 1991: Prevention of the Failure of Metals Under Repeated Stress (by the staff of Battelle Memorial Institute) (New York: John Wiley & Sons, Inc. 1941). Fatigue Design Handbook (SAE Iron and Steel Tech. Comm.) (New York: Soc. of Automotive Engrs., Inc., 1968). S. Suresh, Fatigue of Materials. (Cambridge: Cambridge Univ. Press, 1991).
I am most grateful to Kenneth White, UH Mech. Engr. Dept., for his counsel.

The typical fatigue failure of a crank. (This image and the Comet above are courtesy of Wikipedia)
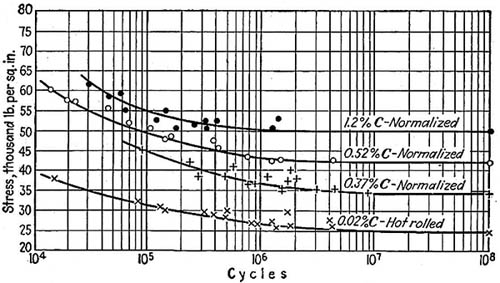
This typical plot of breaking stress vs. no. of cycles (or S-N curve) for four carbon steels is taken from 1921 Univ. of Ill Exp. Sta. Bulleting 124.

Electron microscope image showing several micro-cracks in AL2O3. (From Rühle, Clausen & Heuer, 1986, Copyright American Ceramic Soc.)