Modeling the Atom
by Andrew Boyd
Today, guest scientist Andrew Boyd models the atom. The University of Houston presents this series about the machines that make our civilization run, and the people whose ingenuity created them.
I remember my introduction to the atom in elementary school. I learned that atoms were made of protons, neutrons, and electrons; that protons and neutrons were situated in the middle, or nucleus, of the atom, and that electrons orbited around the nucleus. I made atoms out of Styrofoam balls decorated with paint and glitter. Toothpicks held the nucleus together, and circular strands of copper wire kept the electrons in their orbits. My model of the atom looked a lot like a picture of our solar system.
The earliest scientific model of the atom didn't have protons, neutrons, and electrons. Instead, atoms were modeled as small, indivisible bits of matter. The word atom derives from the Greek word tomos, which means "to cut," and the prefix a, which means "not." The atomos proposed by Democritus in the fifth century B.C. were literally "uncuttable" bits of matter.
This early model of the atom lay dormant until the middle of the eighteenth century,
when scientific experimentation gave new life to atomic theory. Atoms could actually help explain things people were observing, a vital prerequisite for anyone to care about a model. However, it was not until 1897 when J. J. Thomson discovered the electron that atoms became more than uncuttable bits of matter. Thomson's new model had electrons held together by a soupy goo of positive charge to balance the negative charge of the electrons, and it came to be known as the plum pudding model of the atom. In 1910, Ernest Rutherford discovered there was an awful lot of empty space in atoms, leading him to postulate yet a new model of the atom, in which electrons orbit protons and neutrons in the nucleus. In 1913, Neils Bohr changed the model again. Bohr's model looked a lot like Rutherford's, but only allowed the electrons to circle in very special orbits. This was the model we were taught as children.
But the biggest change came with the work of Erwin Schrödinger, who did away with the notion that electrons actually move in orbits around the nucleus. With Schrödinger's model, we stopped thinking about how electrons move and resigned ourselves to thinking about where we'd expect to find them if we went looking. Orbits were replaced by "orbitals," a name that pays due respect to the history of atomic models, but is actually somewhat misleading. Orbitals have nothing to do with orbits, they're just mathematical statements about the probability of finding an electron in a particular place. In fact, in Schrödinger's world, and in the world of quantum mechanics more generally, the whole question of what it means for an electron to get from point A to point B becomes considerably more complicated.
![]()
Schrödinger's model may not be as easy for us to picture as earlier models, but we use it because it explains many experimental observations that the earlier models simply couldn't. Today's model of the atom is typical of an underlying trend in science and engineering. More and more, we find transparent physical models yielding to mathematical abstraction. I'm just having trouble trying to explain our modern view of the atom to my children using Styrofoam, glitter, and copper wire.
I'm Andy Boyd, at the University of Houston, where we're interested in the way inventive minds work.
(Theme music)
Dr. Andrew Boyd is Chief Scientist and Senior Vice President at PROS, a provider of provider of pricing and revenue optimization solutions. Dr. Boyd received his A.B. with Honors at Oberlin College with majors in Mathematics and Economics in 1981, and his Ph.D. in Operations Research from MIT in 1987. Prior to joining PROS, he enjoyed a successful ten year career as a university professor. His new book, The Future of Pricing: How Airline Ticket Pricing Has Inspired a Revolution, (New York: Palgrave-MacMillan), is to come out in October, 2007.
R. E. Dickerson, H. B. Gray, and G. P. Haight, Chemical Principles. 2nd ed. (The Philippines: W. A. Benjamin, 1974).
For more on the quantum atom and its history, see, C-l. Tien and J. H. Lienhard, Statistical Thermodynamics. (New York: Hemisphere Pub. Co., 1976): Chapters 4 through 7. (Schrödinger equation and diatomic molecule model from this source.)
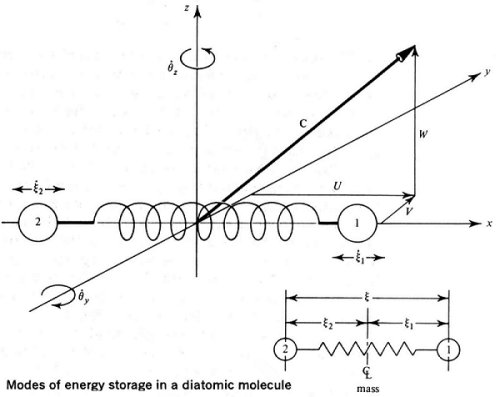
Model of a diatomic molecule suggesting different modes of energy storage, each of which contributes 1/2kT to the molecule's energy (k is Boltzmann's constant and T is absolute temperature.) 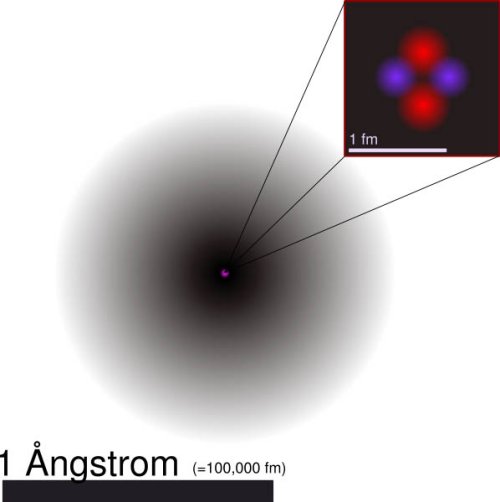
Modern helium atom model. The gray cloud suggests the probability density of the 1s electron. (Image courtesy of Wikipedia)