Greenhouse Gases
Today, a closer look at greenhouse gases. The University of Houston presents this series about the machines that make our civilization run and the people whose ingenuity created them.
We all have some idea of how the greenhouse effect works. Most of the hot sun's energy enters in the short visible wavelengths, and it warms the earth. Then, at night, the warm earth re-radiates its warmth back to outer space. But radiation away from Earth rides on longer wavelengths. And its escape is impeded by many gases in the atmosphere - water vapor, carbon dioxide, methane, ozone, and traces of others. We call those, greenhouse gases.
Earth's temperature will stay in balance if these gases stay constant. Too much of any of those gases, and Earth's temperature will rise. Too little - it will fall. Water is the dominant greenhouse gas. Next is carbon dioxide, then methane and ozone.
Ozone is tricky: It does little to block heat leaving Earth - its greenhouse role is pretty minor. But it protects us by blocking most of the intense ultraviolet radiation from the Sun. We seriously need the ozone sunscreen in the upper atmosphere. At the same time, ozone at Earth's surface is a serious health threat.
So let's look at the big three: water, carbon dioxide (or CO2) and methane. First, water: It constantly cycles: Rain and snow balance evaporation. But there's a catch. If the other greenhouse gases increase in the atmosphere, then Earth's temperature will rise. That, in turn, lets air hold more water in gaseous form, which will increase warming. And we have a vicious circle.
As to the other two: a pound of methane is far more effective in blocking the escape of heat from Earth to outer space than a pound of CO2. The good news is: methane breaks down within a decade or two. CO2 lasts much longer, and there's a lot more of it. The result: All eyes turn toward CO2.
We know some things for sure about CO2 and warming: We know that Earth's average temperature increased about a degree and a half Fahrenheit between 1970 and 2017. We also know that CO2 rose from two to three hundred parts-per-million, four times over the last 400,000 years. The first three times, it then fell back again.
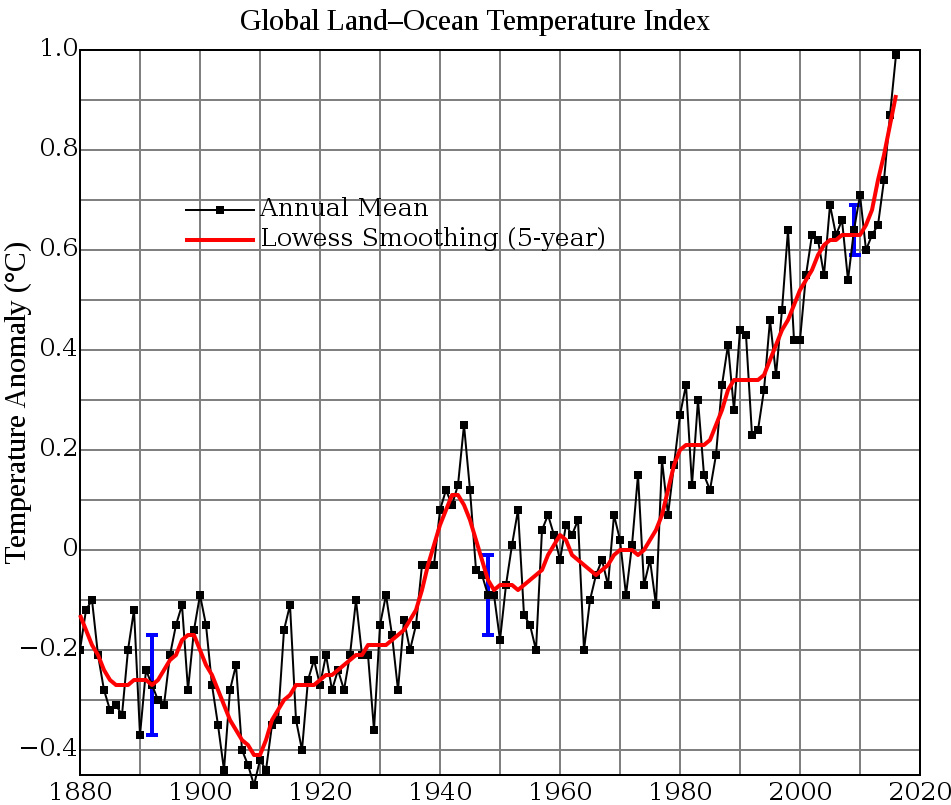
Global temperature change since 1880. Image courtesy NASA Goddard Institute for Space Studies. For more information see https://pubs.giss.nasa.gov/abs/ha00510u.html. Accessed Sept. 3, 2017
But the last rise reached a level of 300 by the beginning of the 19th century - the beginning of the industrial age. Then, instead of falling, it took off in an exponential pattern and rose another hundred parts per million in just two hundred years.
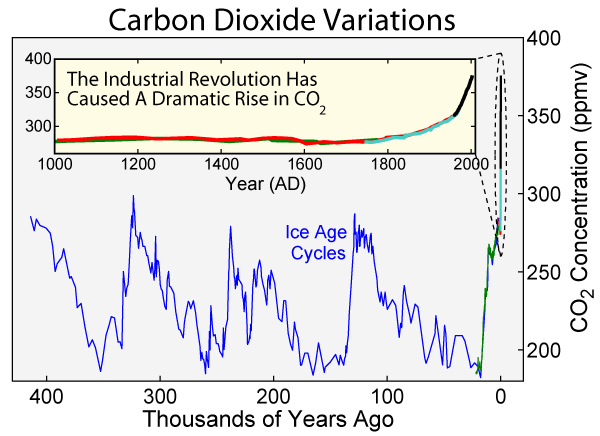
Atmospheric carbon dioxide over the past 400,000 years. (Note the expanded scale for the millennium.) Image courtesy of Wikimedia Commons. For details Click Here.
So: Are human emissions responsible? We presently add over one percent to the total CO2 in the atmosphere each year - some 11 percent per decade. We have here, more than a just a perfect correlation between increased CO2 and temperature rise. We also have a clear mechanism of cause and effect.
The clincher comes in recent studies of isotopes of carbon that plants take from the air. They show, beyond question, that increasing portions of atmospheric CO2 are from fossil fuels. And we're at the point where it's painfully certain that the rise of CO2 and temperature are connected, and the undeniable result of human activity.
I'm John Lienhard at the University of Houston, where we're interested in the way inventive minds work.
See the Wikipedia articles on Greenhouse gas, Carbon dioxide in Earth's atmosphere, and Temperature record.
See also, the 2014 IPCC (Intergovernmental Panel on Climate Change) report. And the full 2016 Environmental Protection Agency's "Climate Change Indicators in the United States" report.
Time did not permit me to go into the detail I would've liked, in the text above. For example, not all water in the atmosphere creates positive feedback to global warming. Water in droplet form (clouds), rather than gaseous, can reflect the sun - the albedo effect - and provide some desirable negative feedback.
Some human contributions, present in amounts much smaller than water and CO2, are also strong global warming agents that add to warming - nitrous oxide and sulfur hexafluoride, for example. And CO2 not only affects the atmosphere. The ocean also absorbs a significant amount of CO2. If it didn't, atmospheric CO2 would now be near 500 instead of 400 ppm. However, the ocean has meanwhile grown more acidic as a result.
Finally, I allude at the end to measurements of lighter isotopes of carbon in the atmosphere. These isotopes appear in higher proportions in fossil fuel CO2. And are now appear in plant up-take of atmospheric CO2. For more on this, see this report and its references.
This episode was first aired on September 8, 2017.