Decompression Sickness
by Janeen Judah
Today, decompression sickness, the bends and Boyle's Law. The University of Houston presents this series about the machines that make our civilization run, and the people whose ingenuity created them.
Decompression Sickness is the modern term for the illnesses caused by a sudden reduction in the pressure around the body, today most commonly in recreational or commercial diving, but also with aviators, astronauts and compressed-air workers. French miners were the first to show the symptoms from breathing pressurized air in a mine in the 1840s, but it became famous in the 1870s during two high profile bridge projects using caissons for their foundations.
What are caissons? They are huge bottomless boxes sunk to a riverbed to build foundations for bridge towers. Water is pumped out of the boxes, and compressed air is pumped in to keep the water out. Workers dig inside the boxes, removing sediment down to bedrock, then fill the caissons with concrete to form the bridge tower foundations.
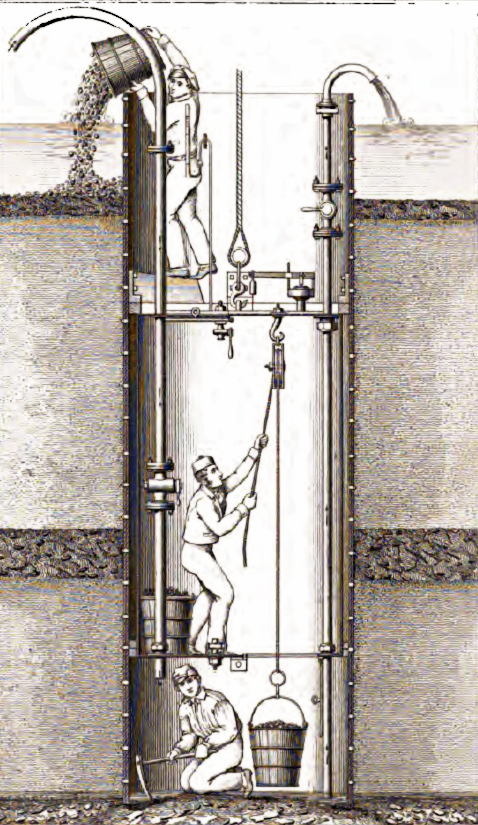
A diagram of an open caisson, devised by Jules Triger, dated 1846. Procédé Triger" by Am'd'e Burat - G'ologie appliqu'e ou Trait' de la recherche et de l'exploitation des Mines. Licensed under Public Domain via Wikimedia Commons.
The Brooklyn Bridge and the Eads Bridge in St Louis used caisson foundations. In both projects, caisson workers developed a mysterious debilitating disease, with pain in the joints, a bent forward posture, blindness and sometimes death.
Scientists discovered that tiny bubbles of nitrogen caused the "caisson disease" or "the bends." Bubbles come out of solution in workers' blood as they decompress from the pressurized air in the caisson to the surface. You can see this for yourself the next time you open a bottle of soda. In a closed bottle, the soda is under pressure and the dissolved gas bubbles are too tiny to see. Opening the bottle reduces the pressure, so the gas bubbles get bigger and foam out.
To understand how the bubbles form, it helps to know Boyle's Law. Sir Robert Boyle was a 17th century Irish scientist — a pioneer of modern chemistry and the scientific method. Boyle did a famous experiment in 1662. He used mercury in a sealed U-tube to discover the effect of an increase in pressure on a set volume of air. Boyle's Law states that a gas volume is inversely proportional to pressure; in other words, at twice the pressure, a gas will have half the volume.1 So, as pressure is reduced by opening a soda bottle, coming up from an underwater dive — or returning to the surface from a pressurized caisson — tiny bubbles in the soda, or bloodstream, get bigger. At half the pressure, twice the volume.
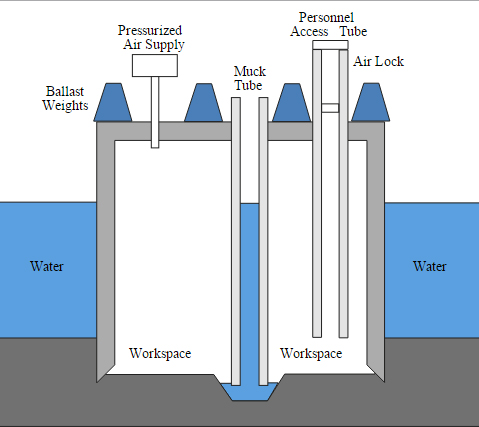
The principal features of a caisson are the workspace, pressurized by an external air supply, and the access tube with an airlock. Photo credit: Yk Times/ Wikipedia
Today, the most common way people breathe compressed air is in recreational or commercial diving. Decompression sickness is prevented by limiting time under pressure and cured by time in a recompression chamber. In recreational diving, limiting underwater time and depth minimizes nitrogen saturation. In commercial diving, divers are monitored carefully underwater, then enter a recompression chamber when they come to the surface. Commercial divers continue to breathe compressed air in the sealed chamber as the air pressure decreases slowly over several hours.
Doctors and dive physiologists are still trying to figure out the effects of dissolved gases in the body. They don't fully understand the disease made famous by the caissons of the Brooklyn Bridge.
I'm Janeen Judah, for the University of Houston, and interested in the way inventive minds work.

Brooklyn Bridge. Photo credit: Wikipedia/ Юкатан
(Theme music)
Endnotes:
On Boyle’s Law: I have quoted the law assuming the constant temperature of the human body. Boyles’ Law is a special case of the ideal gas law, PV = RT and is true only when temperature is constant. P1V1 = P2V2.
Robert Boyle, Boyle’s Law and his famous U-tube (not YouTube) experiment are widely discussed in chemistry and physics textbooks.
Instruction on decompression sickness is an important part of certification courses for recreational divers. For detailed information, see the PADI (Professional Association of Dive Instructors) Encyclopedia of Recreational Diving, 2008. Pp. 5-41 to 5-61.
DAN (Divers Alert Network) is the premier resource for both divers and hyperbaric physicians. DAN’s article on decompression illness can be found here.
For a video of a Navy decompression chamber in action, go here.
Engines episode 2691, The Body at Vacuum, also discussed what happens when ambient pressure is rapidly reduced, but in space.
The episode originally aired on August 21, 2015.