What Lives Within Us
Today, what lives within us. The University of Houston presents this series about the machines that make our civilization run, and the people whose ingenuity created them.
The germ theory of disease was centuries in the making. The idea that tiny, unseen organisms could cause disease was proposed sporadically for over a thousand years, but not refined and accepted until fairly recently. Microbes were first identified by van Leeuwenhoek in the late 17th century, and it was still later in the 1840's when Ignaz Semmelwiez demonstrated the importance of hand washing to prevent surgical infections; John Snow revealed the waterborne origin of cholera in 1854 London; a little later Robert Koch developed the postulates of infectious disease. It became clear that controlling transmission of certain organisms could prevent such illness, and later that certain drugs could cure them. Sanitation methods became our protective curtain, antibiotics our targeted weapons against the lurking threat of infectious disease.

Left: Antonie van Leeuwenhoek invented one of the first practical microscopes. He used these instruments to explore bacterial life and truly opened the world of microbiology.
Right: Drawing of van Leeuwenhoek's microscopes, side view.

Prussian physician Robert Koch identified the organisms responsible for anthrax, TB, and cholera. He also co-developed the postulates of infectious disease, used to determine if a disease can be attributed to a microbial organism.
Bacteria and other microbes are associated more with disease than health, an "us vs. them" mentality. But there is another side to the story. The use of antibiotics may have negative consequences, among them wiping out beneficial organisms along with those causing disease. The human body is in fact colonized with a wide variety of "commensals" — organisms that live in our gut, mouth, nose and on our skin — without causing harm.
We've cultured many of these organisms in the laboratory, but powerful new tools of genetic analysis now make possible the identification of large numbers of previously unrecognized organisms. The body doesn't exist as the simple element we recognize as a species — as us.
Legendary molecular biologist Joshua Lederberg wrote that "We should think of each host and its parasites as a superorganism with the respective genomes yoked into a chimera of sorts." If that sounds farfetched, consider this: 9 of 10 cells in the human body are microbial, not identifiable as human. In the human gut alone, 'as many as 1000 species bring to the body 100 times as many genes as our own DNA carries.' Each of us is a community of living organisms, an ecosystem existing in a concerted life together. Genetically, we are vastly outnumbered by our own microbiome, the term that describes the genetic makeup of our commensals.
The Human Microbiome Project hopes to determine if there is a consistent, identifiable microbiome, and whether changes in this microbiome correlate with changes in health and disease. This is the cusp of a new understanding; whether it will lead to a revolution in health similar to sanitation and antibiotics remains to be seen, but it is most tantalizing. It's likely that a complete picture of a person's state of health must include the general health of their commensals.
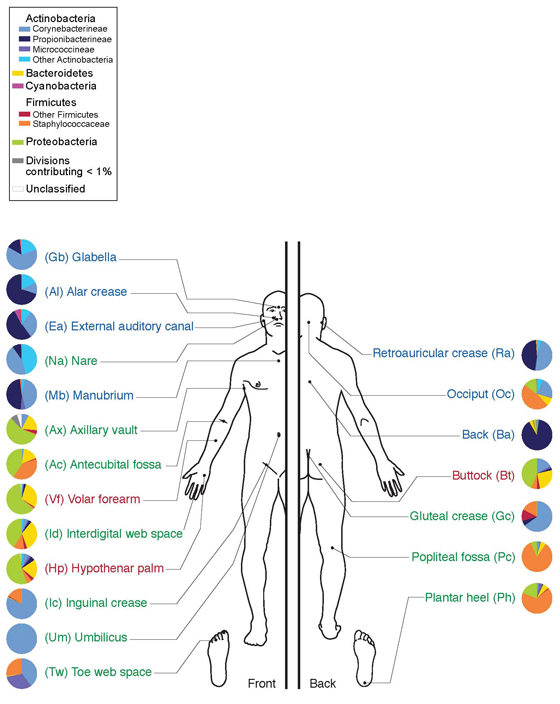
Skin microbiome chart.
This shows the distribution of certain organisms which can now be identified from the skin from various parts of the body. An amazing finding is that each area seems to have a characteristic and possibly unique microbial ecosystem associated with it. The concept of biodiversity takes on a new scope as applied to the microbial residents of the human body.
An interesting question concerns removing the human from the usual environment and moving to somewhere more artificial, more "sterile", such as a spacecraft. Which of these commensals should go along to ensure health and development? Which might we want to leave behind, wary that they may turn on us and cause an opportunistic infection at the first sign of immune weakness?
At least two things are clear. We are never really alone, and we as superorganisms need to get to know our microbes in residence.
I'm NASA astronaut Michael Barratt for the University of Houston, where we're interested in the way inventive minds work.
(Theme music)
End Notes:
It has long been known that certain living organisms inhabit bodies of more complex creature hosts, such as us. There are many terms for these organisms, which may be classified based on the relationship between them and their host. A commensal is an organism that coexists with its host in a non-harmful way. We think of a parasite as coexisting with but causing harm to its host, while a mutual both delivers benefit to and receives benefit from its host. For the human body, there is an equilibrium depending on the number and type of these organisms, surrounding conditions, diet, immune health, and other factors that may alter an organism's relationship with its host.
Many areas of the human body are colonized with organisms. As our outermost layer, the skin harbors a variety of bacteria, most of which are harmless as long as the skin is intact. The human gut is a veritable crucible of life. This is not surprising when one considers it as a vessel filled and continuously replenished with nutrients, moisture, and warmed to a pleasant temperature. This is an obvious area of focus for studying the biodiversity of the body. We have been able to identify certain bacteria, viruses, and other organisms using laboratory culture techniques, but these are limited to the organisms that are amenable to growth in this artificial medium. The powerful new techniques of genetic analysis, and in particular metagenomic analysis, allow us to identify the genetic character of whole populations of organisms in a community. As a result, we can realize the true extent of biodiversity in a certain region of the skin or the gut. With regard to our bacterial and viral commensals, we simply didn't know what we were missing. This is most definitely an area to watch.
Electron microscope image of a common bacterial nemesis, Escherichia coli.
References:
Shining a Light on the Genome's Dark Matter. Elizabeth Pennisi Science 17 December, 2010; page 1619.
This is a nifty, brief overview which gives a simple of description of the Human Microbiome Project. It cites some of the impressive numbers of relative populations of human cells and DNA to their microbial counterparts.
The Human Microbiome Project
The National Institutes of Health Human Microbiome Project provides a quite user friendly explanation of the project and its scope in understanding the human superorganism.
Honor thy Symbionts, by Xu and Gordon.
For those who want to know more and have a familiarity with biological terminology, Ji an Xu and Jeffrey Gordon have written a fine article describing the relationship between host and bacterial symbionts. Aside from some detailed descriptions of some of the organisms inhabiting the human gut, there is very good overall discussion of how the human relates to the microbial world in general.
Images
All but the NIH diagram of the Human Body and its commensal are from the Wikimedia Commons. The former is in the public domain as having originated from a federal institute.