James Dewar
Today, the man who froze hydrogen. The University of Houston's College of Engineering presents this series about the machines that make our civilization run, and the people whose ingenuity created them.
 The thing to know about James Dewar at the outset is that he took up violin-making in 1852. He was a ten-year-old Scottish boy who'd just come down with rheumatic fever. That was long before antibiotics, or even the over-counter medicine-of-choice, aspirin. During his two-year convalescence from the fever's crippling effects, he began learning the complex craft of making violins.
The thing to know about James Dewar at the outset is that he took up violin-making in 1852. He was a ten-year-old Scottish boy who'd just come down with rheumatic fever. That was long before antibiotics, or even the over-counter medicine-of-choice, aspirin. During his two-year convalescence from the fever's crippling effects, he began learning the complex craft of making violins.
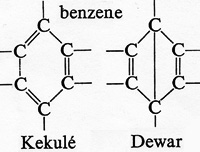
Dewar went on to study chemistry at Edinburgh, and did very well. He was a Cambridge professor at the age of 33. By then, he'd done many things -- including work with Friederich Kekulé on the still-puzzling compound, benzene.
It was Kekulé who'd realized that benzene's six carbon and six hydrogen atoms had to combine in a hexagonal ring. But Dewar found other possible arrangements, based on what we then knew about chemical bonds. So why didn't benzene take those other forms as well? The answer didn't emerge until we had quantum mechanics. But Dewar had shown the need for a deeper understanding than chemistry could yet provide.
 That kind of meddlesome curiosity is what drives new knowledge and it certainly drove Dewar. He was into everything. He helped invent the smokeless powder, cordite. He measured the temperature of the sun and the temperature of an electric spark. He did a lot of important early spectroscopy. He studied bubble behavior. And his name lives today, not from any of that, but for the dewar flask -- what has today become he familiar thermos bottle.
That kind of meddlesome curiosity is what drives new knowledge and it certainly drove Dewar. He was into everything. He helped invent the smokeless powder, cordite. He measured the temperature of the sun and the temperature of an electric spark. He did a lot of important early spectroscopy. He studied bubble behavior. And his name lives today, not from any of that, but for the dewar flask -- what has today become he familiar thermos bottle.
When Dewar began studying low-temperature material behavior, he was drawn to the problem of condensing gases into liquid. By the late 19th century, every gas but hydrogen and helium had been liquefied. Helium had yet to be discovered, and no one had any idea just how cold hydrogen would have to get before it condensed.
I won't go into Dewar's long struggle. But he finally got below -252oC and succeeded. And he didn't stop there. He got down within 13 degrees of absolute zero and froze hydrogen solid. Next, he turned to the new gas helium. But the Dutch condensed it first.
His flask, crucial in that work, used a silvered, evacuated space in a double wall to block heat flow; but how to make the vacuum low enough? Dewar found that charcoal eats gas at low temperatures. So he placed a bit of charcoal in the gap; then evacuated it as best he could. When cold liquid gas filled the tank, the charcoal removed the remaining air from the wall space, and made the insulation nearly perfect.
And we're back to a young boy making violins. That's where James Dewar honed his uncanny laboratory skills. Take someone with his ability to think abstractly, combine that with the patient care needed to build something that really works, and the result is formidable. It is something we all might well reflect upon as we pour hot coffee or cold lemonade from our next picnic thermos bottle.
I'm John Lienhard at the University of Houston, where we're interested in the way inventive minds work.
A. B. Costa, Dewar, James. Dictionary of Scientific Biography, C. C. Gillispie, ed., Vol. IV (New York: Charles Scribner's Sons, 1975): pp. 78-82.
See also the Wikipedia articles on James Dewar and vacuum flasks.
My thanks to colleagues Chengzhi Cai (UH Chemistry Dept.), and Ken White (UH Mech. Engr. Dept.), and David Perkins for counsel and assistance. Dewar image courtesy of Wikipedia Commons, dewar flask photos by J. Lienhard.
