Lazare & Sadie Carnot
Today, meet Lazare and Sadi Carnot. The University of Houston's College of Engineering presents this series about the machines that make our civilization run, and the people whose ingenuity created them.
Lazare Carnot was a remarkable eighteenth century figure, even though his son and grandson are better known. Son Sadi Carnot led us to the Second Law of Thermodynamics. Grandson Sadi Carnot became President of France.
Born in 1753, Lazare studied math and military engineering. During several years of lackluster military service, he competed for math prizes and became involved with brewing French Revolution politics. Then he got himself thrown in jail for an overzealous affair with an aristocrat's daughter.
He might've languished there but, as the Revolution began, he was pulled from prison, back into service. Seven years later, he was made a member of the five-man Directory that now ruled post-revolutionary France. In that role, Carnot started Napoleon's rapid rise by giving him charge of an Army. As long as Napoleon reigned, Carnot did high-level government work. But afterward, the returning monarchy remembered Carnot's vote to behead the king. So he had to spend the rest of his life exiled in Germany.
Lazare Carnot was very multitalented. He was an excellent violinist and he was strongly interested in technology. He be-friended people like the Montgolfier Brothers, and Robert Fulton who showed up in France, trying to sell submarine designs. At his core, Carnot thought like a technocrat. He once wrote,
if real mathematicians were to take up economics and apply experimental methods, a new science would be created. [It] would only need to be animated by the love of humanity ... to transform government.
The eighteenth-century French aristocracy had embraced Newton over his rival Leibniz. But Carnot came down on the other side of that fence. He not only used Leibniz's calculus, he also made fine use of Leibniz's ideas about mechanical energy conservation.
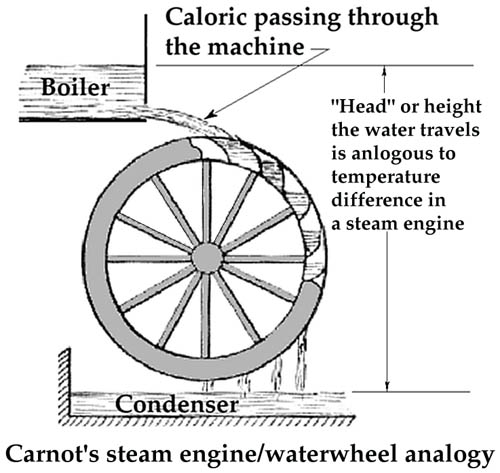
Example: he pointed out that, in an imaginary perfect waterwheel, none of the water's energy would go to waste. None would be dissipated, and all the motion would be completely reversible. If the perfect waterwheel were run backward, it would become the perfect pump. And here Lazare's son Sadi claimed his inheritance.
He said, let us conceive a perfectly reversible steam engine. If we could build such a thing, we could run it in reverse and pump heat from a condenser to a boiler. Refrigerators wouldn't appear until thirty-six years later, but Sadi Carnot had pointed the way.
He proved that no engine could do better than that imaginary engine; and he wrote Carnot's Theorem. It said that the power an engine delivers depends on the high temperature (in the boiler) and the low temperature (in the condenser), just as a waterwheel depends entirely on how far water falls through it. That was where Sadi Carnot put us on the road to the Second Law of Thermodynamics.
So an extraordinary father and son provide a remarkable example of both revolution and continuity. They remind us that, to make a great leap, a mind must be revolutionary. But they also remind us that nothing comes out of nothing.
I'm John Lienhard, at the University of Houston, where we're interested in the way inventive minds work.
For material on Lazare Carnot, see C. C. Gillispie, Lazare Carnot Savant. Princeton, NJ: Princeton University Press, 1971), or C. C. Gillispie, Carnot, Lazare Nicolas-Marguerite, Dictionary of Scientific Biography. Vol. III (C.C. Gilespie, ed.) (Chas. Scribner's Sons, 1971).
For material on Sadi Carnot, see J. F. Challey, Carnot, Nicolas Léonard Sadi, Dictionary of Scientific Biography. Vol. III (C.C. Gilespie, ed.) (Chas. Scribner's Sons, 1971), or read Mendoza's Introduction in S. Carnot, Reflections on the Motive Power of Fire, (ed. E. Mendoza) (Gloucester, MA, Peter Smith, 1977.)
For more on the second law, your best bet is one of the older engineering thermodynamics textbooks. I recommend J. H. Keenan, Thermodynamics.New York: John Wiley and Sons, Inc., 1941 and further editions. (You will, however, have trouble trying to find a book that clearly says where Carnot stops and actual statements of the 2nd law begin.)