Temperature
Today, we ask what temperature is. The University of Houston's College of Engineering presents this series about the machines that make our civilization run, and the people whose ingenuity created them.
Very early in any thermodynamics class, I ask students to tell me what they think a thermometer measures. Think about that: A thermometer gives us a number that we call temperature, but what have we really measured? All the reasonable answers seem to crumble in our hands when we try to hold them in the light.
One student asks, "Isn't temperature a measure of heat?" That sounds reasonable, but suppose we're boiling water. We heat it and heat it until we've turned all the water into steam. But now the steam is the same temperature the water was. All that heating and no temperature change.
Another student asks, "Doesn't temperature measure the speed of molecules?" That turns out to be true as long as the molecules form a very thin gas. It doesn't work at all for complicated materials.
So take another tack. Maybe temperature just measures the length of a mercury or alcohol column. Okay, suppose we fill a capillary tube with mercury or alcohol, dip it in a glass of ice water, draw a line, and call it zero degrees Celsius. Then we dip it in boiling water, and we mark off a hundred degrees Celsius.
Finally, we ask where we should mark off fifty degrees Celsius. The answer is not right in the middle of the column. Neither mercury nor alcohol expands exactly the same amount for each degree of temperature change. The two thermometers would read a little bit differently. We need some independent means for finding out where the fifty-degree mark would fall.
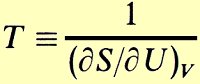 Not until halfway through the course can we finally define temperature so all the many pieces fit together. And when we do, the definition is too technical for me to say on the air.
Not until halfway through the course can we finally define temperature so all the many pieces fit together. And when we do, the definition is too technical for me to say on the air.
Voice: No, wait a minute. You can't leave us hanging.Really, you don't want to know.
Voice: Sure we do. Tell us.
Okay, okay. "The temperature of any material is the partial derivative of its energy with respect to its entropy while you hold its volume constant."
Voice: You're joking!
No, I told you it'd sound arcane. And yet, science has to be about making sense. In the end, our senses tell us what temperature is, at least between the freezing point of our bodies and about 140 degrees Fahrenheit, where our nerves begin to burn out.
Temperature is a measure of intensity. The higher the temperature of anything, the greater that intensity is, and the more heat it will drive into colder bodies. Temperature can really be thought of as a measure of the ability to drive a flow of heat.
And I'm reminded once more that, objective as we try to be, our subjective selves always inform science. Let me leave you with a fine insight into temperature and intensity by poet Christopher Morley:
I bid you, mock not Eros;
He knows not doubt or shame,
And, unaware of proverbs,
The burnt child craves the flame.
I'm John Lienhard, at the University of Houston, where we're interested in the way inventive minds work.
(Theme music)
For proper discussion of temperature, see any undergraduate thermodynamics text. I especially recommend W. C. Reynolds, and H. C. Perkins, Engineering Thermodynamics. Garden City, NJ: Doubleday and Co., Inc., 1966.
For a discussion of temperature in relation to molecular velocity, see, e.g., C. L. Tien, and J. H. Lienhard, Statistical Thermodynamics. Revised ed. Washington, D.C.: Hemisphere, 1979.
The Christopher Morely quotation is from his poem, Of a Child That Had a Fever.
