Energy is Pure Delight
Today, we chase a will-o-the-wisp. The University of Houston's College of Engineering presents this series about the machines that make our civilization run, and the people whose ingenuity created them.
Energy is pure delight, wrote William Blake. But for me the purest delight of energy is its elusiveness. Ask the dictionary what energy is. It says it's "the capacity to do work." Then it tells you work is a transference of energy. The technical literature takes you around the same circle. For energy is a primary concept, like force or space. We cannot define it. We can only compare experience and then try to agree upon that experience.
The binding tissue around energy is a physical principle: the Conservation of Energy or the First Law of Thermodynamics. We know forms of energy can be swapped. We work at rubbing our cold hands together and they grow warm. I run electricity through a light bulb. The electricity is transformed. It leaves as light and heat.
We take those equivalences for granted. But we had no light bulbs three centuries ago: no electricity, no steam engines. In the late 18th century we decided heat must be an indestructible material called caloric. When a carriage wheel heated its axle, we thought the rubbing was releasing caloric from the wood. It never occurred to anyone that energy was being transmuted from one form to another.
But the new 18th-century steam engines undermined that thinking. Steam power plants burned coal, boiled water, and let the steam's energy do work. For a while, people tried to compare steam engines with water wheels. Water flows through a water wheel, does work, and degrades gravitational energy. Shouldn't steam engines work the same way, with caloric flowing through and being conserved the same way water is? No one saw that heat was not being conserved but was being transformed into work.
In the 1790s Count Rumford, an American expatriate working in the Bavarian court, took an interest in artillery. Rumford noticed that when cannon barrels were reamed they got much hotter if the bit was blunt. Instead of cutting, there was a lot of frictional rubbing. Rumford found he could keep generating heat as long as he worked at turning the bit. So much for releasing caloric (or heat) from the surface! The supply of surface caloric in the material would have to be inexhaustible to do that.
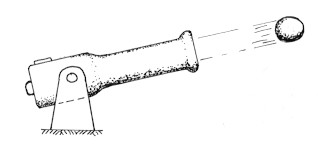
Rumford did another, very different, cannon experiment. He fired cannons with and without cannonballs. The cannons without cannonballs were far hotter after they'd been fired. Why? The reason was, the explosion did work accelerating the cannonball. The ball carried energy away. When there was no cannonball, the energy couldn't change form, and it simply heated the cannon.
So, piece by piece, a concept took on flesh and blood. Energy is pure delight just because its form keeps changing. But that concept is subtle and difficult. A hundred and fifty years ago, we didn't have it sorted out. And today our dictionaries still don't know what to do with energy -- elusive, changing, and ever-present.
I'm John Lienhard, at the University of Houston, where we're interested in the way inventive minds work.
(Theme music)
These matters are discussed in any undergraduate engineering thermodynamics text, of which there are dozens. My recommendation would be: Reynolds, W. C. and Perkins, H. C., Engineering Thermodynamics. New York: McGraw-Hill, Inc., 1970, 1977.
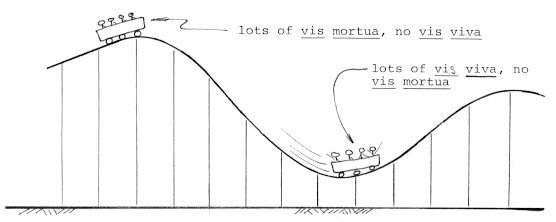
Restrictive illustration of the law of Conservation of Energy: Roller coasters exchange two forms of mechanical energy. Potential energy (vis mortua) becomes kinetic energy (vis viva) which becomes potential energy again. Since roller coasters also gradually dissipate mechanical energy, each successive hill has to be lower.